2021 Faculty of Medicine Distinguished Achievement Award: Torsten O. Nielsen!
We would like to give a warm congratulations to our very own Torsten O. Nielsen for receiving the University of British Columbia’s 2021 Faculty of Medicine Distinguished Achievement Award: Excellence in Clinical or Applied Research. This award recognizes faculty members who exemplify outstanding clinical or applied research and scholarly contributions, and who are dedicated to advancing both the Faculty’s values and vision of transforming health for everyone. We are proud of you, Torsten!
For a curation of publications by the man, the myth, the legend himself, please visit: https://scholar.google.ca/citations?user=2jTP-TEAAAAJ&hl=en.

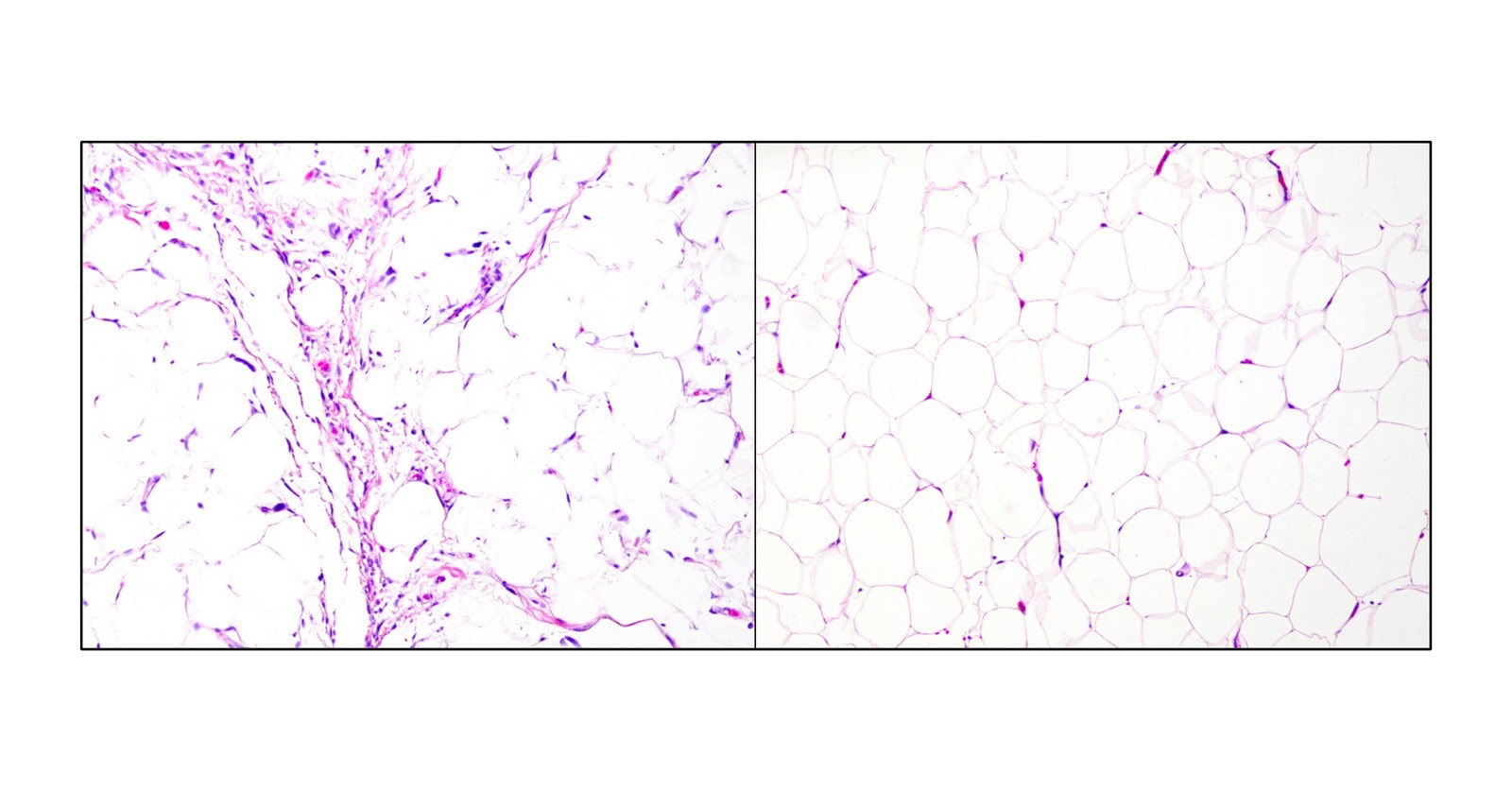
New test provides fast and accurate diagnosis of liposarcomas
Researchers have leveraged the latest advances in RNA technology and machine learning methods to develop a gene panel test that allows for highly accurate diagnosis of the most common types of liposarcoma. It quickly and reliably distinguishes benign lipomas from liposarcomas and can be performed in laboratories at a lower cost than current “gold standard” tests. The new assay is described in The Journal of Molecular Diagnostics.
Spring Social: Tactical Laser Tag at Nu-Zone

Summer Social: Dr Torsten Nielsen’s Birthday Party


Congratulations to Amanda Dancsok for her successful PhD defence!

Congratulations to Samantha Burugu for her successful PhD defence!

Congratulations to Jamie Yu on her successful PhD defence!

Terry Fox Run

Summer Social: Picnic at Cates Park

Winter Social: Curling

Congratulations to Aimee Laporte for her successful PhD defence!

Spring Social: Snowshoeing at Cypress Provincial Park


Summer Social: Pitch & Putt



Dr. Torsten Nielsen @ The 20th Annual Canadian Cancer Society Daffodil Ball
The 20th Annual Canadian Cancer Society Daffodil Ball raised $1.3 million on Saturday, April 16, 2016 in support of Dr. Torsten Nielsen’s critical research in synovial sarcoma.
Winter Social: Bowling

Ki67-QC calibrator available to public
The Ki67-QC calibrator is a web-based calibration exercise designed to calibrate pathologists to a prescribed scoring method. Nine “training” and nine “test” tissue microarray cases representing a wide range of Ki67 scores were chosen to serve as the calibration cases.
This website interface allows pathologists to score the web-image cores via computer mouse click on individual cells. The website software tracks which cells were scored, where those cells were located within the core, whether each cell was scored positive or negative, and the overall Ki67 score for each core. Images of H&E-stained sections from the cores were also uploaded for reference.
The Ki67-QC calibrator is available at: http://www.gpec.ubc.ca/calibrator
NanoString wins FDA approval of breast cancer diagnostic test
GPEC has been working closely with NanoString for the past three years on this FDA approval submission. The FDA approved Prosigna test is based on the breast cancer intrinsic subtype gene expression profiling (PAM50) research, a collaborative study between University of North Carolina, Washington University, University of Utah and GPEC.
More information can be found at:
Summer Social: Hiking at Eagle Bluffs

GPEC publication, second most cited on CCR as of June 1st, 2013
GPEC publication Nielsen et al. CCR 2004 is the second most cited publication on Clinical Cancer Research as of June 1st, 2013. Congratulations to all the authors on this paper!